Yttrium
What is Yttrium?
Yttrium is a silvery metallic transition metal. It is the 39th element in the periodic table. It has similar chemical properties to lanthanides (elements 57-71 in the periodic table) and likewise, yttrium is also classified as a rare-earth element. Rare-earth elements are heavier than iron and are found in low concentrations in the environment. Yttrium is never found as a pure element on Earth, and it only has one naturally occurring isotope. It is used in electronics, synthetic crystals, alloys, and radiation therapy.
Yttrium’s Place in the Periodic Table
Yttrium is in group 3 and period 5. Group 3 elements are similar to lanthanides, and include scandium, yttrium, lanthanum, and actinium. Yttrium is named after a Swedish village, Ytterby, where a mineral containing yttrium was first discovered.
- Atomic number: 39
- Atomic Radius: 180 picometers
- Atomic mass: 88.91
- Symbol: Y
- Group: 3
- Period: 5
- Number of Protons: 39
- Number of Electrons: 39
- Number of Neutrons: 50
- Number of Isotopes: 1 natural isotope
Properties of Yttrium

Yttrium is found on Earth in ores of other rare earth elements such as lanthanides. It is an intermediate conductor of heat and electricity. For example, it has an electrical conductivity 50-times less than copper, but about 2-times the conductivity of nichrome, which is an alloy of nickel and chromium with very high electrical resistance. Yttrium is paramagnetic, which means that it is attracted by magnetic fields. There is no biological role for yttrium, and it is highly toxic.
Physical Properties
Yttrium is solid at room temperature and has a density similar to titanium. It melts at 1526°C, which is similar to iron. It has a boiling point of 3338°C.
- Melting Point: 1526°C.
- Boiling Point: 3338°C.
- Density of Solid Yttrium: 472 g cm-3
- Phase at Room Temperature: solid
Chemical Properties
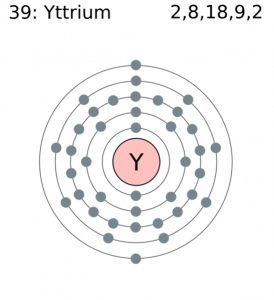
Yttrium has three positive oxidation states, and its oxides are weak bases. It has 3 valence electrons. One electron is in the 4d orbital, leaving space for 9 more. The other two valence electrons completely fill the 5s orbital. Therefore, donation of 3 electrons gives an electron configuration equal to krypton, and the most stable oxidation state of +3. The +3 oxidation state is most common, and is present in oxides, halogens, and other inorganic compounds of yttrium.
However, it can also form compounds with +1 and +2 oxidation states. Yttrium can react with water. However, it is stable in weak acids. It can form binary compounds with carbon, phosphorus, selenium, silicon, and sulfur.
- Oxidation states: +1, +2, +3
- Specific Heat: 0.2 J g-1K-1
- Electronegativity: 1.22 (Pauling scale)
- Heat of Fusion: 17.49 kj mol-1
- Heat of Vaporization: 114.1 kj mol-1
- Electron Configuration: [Kr] 4d1 5s2
Isotopes
Yttrium only has one natural isotope that is stable. Unstable, radioactive isotopes yttrium are produced by nuclear fission. Nuclear fission is a process that leads to the loss of subatomic particles, or radiation, like protons, neutrons, and electrons. Fission of uranium can produce yttrium isotopes Y-90 and Y-91. These have half-lives on the order of days, so they do not accumulate to provide a source of natural isotopes on Earth. More than 30 synthetic isotopes of yttrium have been identified, but they all have half-lives of less than one day.
Alloys and Allotropes
Yttrium is used to enhance the properties of some alloys. As little as 0.1% percent of yttrium can make up alloys of chromium, molybdenum, titanium, and zirconium alloys. This reduces the “grain size” of the metal. Grain size refers to the number of places in the metal where liquid transitions to solid during cooling. Fast cooling, or the addition of yttrium, creates a smaller grain size, which produces a harder and stronger alloy.
Compounds of Yttrium
Yttrium oxide and yttrium oxide sulfide are used to form a structural lattice that is then filled with europium(III) ions. These ions act as a phosphor, and together with yttrium were used to create the first televisions. Yttrium oxide has a high melting point, and is added to some glass and ceramics to prevent them from cracking during quick changes in temperature (e.g. adding hot water to cold glass). Yttrium phosphide is used as a semiconductor in high power lasers.
Interesting Facts about Yttrium
- Samples of rock from the moon that were brought to Earth by the Apollo missions identified yttrium at much higher concentrations than Earth. There is about 30 ppm of yttrium in Earth’s crust, and over 200 ppm on the moon.
- Crystals of yttrium and aluminum are used to make imitation diamonds for jewelry.
- High performance spark plugs in combustion engines are coated with yttrium.
Occurrence and Abundance of Yttrium
Yttrium makes up .0029 % of the Earth’s crust. That is similar to cobalt and scandium, and over 400 times more abundant than silver. In the universe as a whole, it makes up 7 x 10-7 % of the universe. Yttrium is most often found with other rare-earth metals. Minerals that contain sources of yttrium include bastnasite, monzanite, and xenotime (pictured). Pure yttrium can be isolated from these sources by dissolving the rare-earth metals oxides with a strong acid, and conducting a series of chemical reactions that result in the precipitation of pure yttrium. Yttrium is produced in Australia, Brazil, China, India, and the United States. Yttrium is present in human tissue but has no known biological role.
Uses of Yttrium
Most Notable Uses in General
The first color televisions, known as cathode ray tubes, used yttrium to capture electrical signals that could then be converted to colored light by phosphors. Phosphors are materials that emit light when excited by electrons or photons. Yttrium was used to produce red and green light. Common plastics are produced by the joining individual molecules, such as ethylene (C2H4), into long chains, such as polyethylene. This process is known as polymerization, and yttrium is used as a catalyst to initiate the chemical reaction of polymerization. Catalysts are chemicals that reduce the energy required to break bonds during chemical reactions. Crystals that contain yttrium are used to produce white LEDs, as well as strong lasers that can cut metal.
Most Notable Uses in Science
Radioactive isotopes of yttrium are used to treat cancer. Rather than treating the whole body with radiation, the yttrium atoms coat the surface of tiny beads, smaller than a single cell. The beads are also coated with antibodies that allow them to stick specifically to cancer cells. When injected into the body, the beads circulate and adhere to cancer cells. This delivers the radiation therapy directly to the cancer cells, which reduces the toxic side-effects of cancer treatment.
Discovery of Yttrium
Yttrium is named after a Swedish village called Ytterbite. In 1787, a heavy black rock was discovered by Carl Axel Arrhenius in that village. He thought it was a new mineral, so he sent a sample to chemist Johan Gadolin, who identified a component of the rock as yttrium oxide. This oxide was then named yttria. However, yttria contained multiple elements that were all named after the village, Ytterbite: ytterbium, terbium, and erbium.
Yttrium in the Future
Yttrium is being actively researched for uses as a semiconductor. Semiconductors are highly efficient conductors of electricity. However, one challenge with their use is that they have to be kept very cold and under high pressure to maintain their conductive properties. Yttrium hydride was recently shown to retain these properties close to 0°C. However, this required the application of pressure 100,000-times the atmospheric pressure at sea-level. That’s because yttrium oxide is a volatile gas, and there are only two ways of converting a gas into a solid: increasing the pressure or decreasing the temperature.
